From Requirements to Approval - Accelerate Medical Device Development by 30%
.avif)

Achieving Faster CE Mark Approvals with Aligned Elements
You can download the guide directly here.

Measurable Results
with Aligned Elements
increased DHF confidence
Why MedTech Companies Choose Aligned Elements
Purpose-built for medical device ALM - not a generic tool adapted with compliance add-ons.
Requirements Management with Audit Trail
Capture and control requirements with complete, chronological tracking.
Every change is documented, stakeholders informed automatically.
Templates configured to your specific QMS enable setup within days.
Requirements flow seamlessly into risks, tests, and documentation without manual linking.
Verification and Validation Management
Plan, execute, and document verification and validation within the same platform as requirements and risks.
Test cases link directly to requirements and risk controls.
When requirements change, affected tests are automatically flagged for re-execution.
Test results flow directly into your DHF with complete traceability.
Integration with Development Tools
Pre-built connectors integrate Aligned Elements with issue tracking, version control, and test automation systems.
Changes in development tools trigger automatic traceability updates.
Engineers work in familiar tools while Aligned Elements maintains compliance documentation automatically.
.avif)

Key ALM Factors for Medical Device Compliance
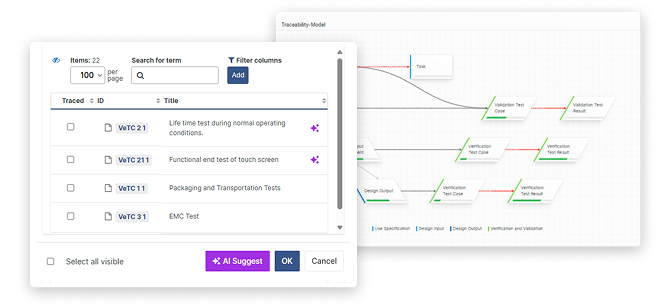
Live AI-driven Traceability
Design Control traceability is the glue that holds your Technical File together. With thousands of traces to manage, manual approaches using Excel matrices become unmaintainable.
A designated ALM provides rule-based, AI-driven traceability management with automatic, real-time gap monitoring eliminating manual merging across multiple systems, all set up according to your own QMS rules.

Integrated ISO 14971 Risk Assessments
Risk drives design. Design drives risk.
A stand-alone risk assessment approach is therefore doomed to fail. Only with integrated risk assessment capabilities spanning from identification, evaluation, mitigation, implementation to verification can real compliance be achieved.
No separate risk management tool, no manual synchronization, no version conflicts.
Real-Time Consistency Checks -
Find Gaps Before Audits
Automated, real-time consistency monitoring finds gaps and errors in seconds. The ALM continuously scans the Technical File for missing risk controls, unreviewed requirements, and broken traceability links.
Teams can now achieve regulatory readiness by addressing compliance issues up-front, not during last-minute audit preparation.


Integration With Existing
Engineering Tools
No system is an island. The power of an ALM can be multiplied by integrating it with existing engineering tool, expanding the data set on which it can operate and thereby eliminating manual handoffs.
Without seamless integration with existing tools, the organization runs the risk of engineers maintaining parallel documentation streams, condemning the documentation to version conflicts and traceability gaps.
Use Case 1: Biotronik Vascular Intervention –
From Duplicate Work to Integrated Design Control Management
Challenge
The Biotronik Vascular Intervention team struggled to create global technical files due to disconnected software tools and redundant Word reports, making compliance tedious and error-prone.
Manual tracking to maintain consistency consumed valuable time, overloading risk management and design control teams leading to user dissatisfaction.
Solution
A dedicated ALM software to unify design control and risk management, reduce redundancies, and support their entire development lifecycle while remaining adaptable to their specific processes was needed.
Selecting Aligned Elements for its configurability and industry-focused design, allowed the team to manage design, test, and risk data in an integrated, consistent way. Tailoring workflows to their needs and leverage interlinked process data lead to greater efficiency and control.
Results
New users quickly found their way in the new an easy-to-use ALM. The integration of requirements, risks, and tests into a single system eliminated redundancies and improved consistency.
The team efficiently migrated legacy data into Aligned Elements and now leverages linked projects for significant data reuse, achieving high data integrity, automated change control, and a complete, consistent audit record across the entire product lifecycle.
Use Case 2: Tecan–
Reducing Documentation Duplication with 80%
Challenge
Tecan’s modular platform strategy enables efficient product development and reuse of hardware components, but regulatory documentation had become a growing bottleneck.
Each product required a separate Design History File even though the same underlying hardware modules were re-used in multiple products, leading to significant duplication of documentation.
Solution
Instead of maintaining a monolithic Technical File per product, Tecan used the Aligned Elements ALM to structure documentation around modular design control packages, each containing specifications, risks, and verifications with full traceability and audit trails.
Product specific Design Control content was separated from module data and linked across modules, enabling controlled reuse on a massive scale, cross-module traceability, and a significantly more efficient Technical File management.
Results
Tecan significantly increased efficiency using a new modular documentation approach. It was further optimized with a shared requirement pool, eliminating duplicate requirements across products.
The change enabled rapid setup of new product documentation within hours by reusing existing, fully traced technical file content.
Implementation Checklist
Phase 2:
Validate the approach using a Pilot Project
Run a pilot project using a new and/or small project to allow team members active during difference phases of the product life cycle to gain familiarity with the ALM.
Gather and analyse the use experience from all team members. Adapt configurations, workflows, templates and process steps to maximize re-use and minimize manual, repetitive tasks in order for each team member to operate as efficient as possible.
Phase 3:
Train users on both the mechanics of the ALM tool and the organizations foreseen use of the tool with focus on the underlying design control concepts. With the result from a tried and tested pilot project, with all kinks ironed out, it is time to roll out broadly. Continuously monitor system usage and compliance indicators to properly integrate the ALM into daily work.
Phase 4:
Establish a proactive governance stance
Pay attention to user feedback as new projects, potentially using new technologies, might require a new or adapted definition of the desired compliance end state.
Update the ALM if new geographical markets are entered. Keep an eye on global regulatory changes (regulations, standards and guidance) and adapt the ALM as new regulatory requirements emerge.
Phase 1:
Define the Target Design Control Model
Start by clearly defining the desired regulatory end state by analysing the existing QMS and development process.
The end state includes the required design control elements, document structures such as the Technical File or Design History File, traceability expectations, and applicable standards and regulations.
Agree on what “complete and compliant” means for requirements, risks, verification, and change control.
Run a pilot project using a new and/or small project to allow team members active during difference phases of the product life cycle gain familiarity with the ALM. Gather and analyse the use experience from all team members. Adapt configurations, workflows, templates and process steps to maximize re-use and minimize manual, repetitive tasks in order for each team member to be as efficient as possible.
Phase 3:
Train and Roll Out
Train users on both the mechanics of the ALM tool and the organization's foreseen use of the tool with focus on the underlying design control concepts.
With the result from a tried and tested pilot project, with all kinks ironed out, it is time to roll out broadly. Continuously monitor system usage and compliance indicators to properly integrate the ALM into daily work.
Phase 4:
Establish a proactive governance stance
Pay attention to user feedback as new projects, potentially using new technologies, might need a new or adapted definition of the desired compliance end state. Update the ALM if new geographical markets are entered. Keep an eye on global regulatory changes (regulations, standards and guidance) and adapt the ALM as new regulatory requirements emerge.
Customer Testimonials





Aligned Elements transformed our audit preparation from a 3-week nightmare into a 3-day routine. The automatic traceability works exactly how we as a QA team think - no more compromises because of generic PLM tools.





With over 1,500 requirements, our Excel-based system was completely at its limit. Aligned Elements gives us the traceability overview and audit confidence we need for FDA and Notified Body inspections. Our last audit: Zero findings due to missing traces.





What convinced us: No marketing fluff, but direct answers from the founder team. Anders and Robert were on a call the next day after our demo and understood our specific GXP requirements. This personal technical expertise makes the difference.





We use Aligned for both risk management (dFMEA 60812 and risk analysis 14971) and requirements management. It is a no nonsense tool, with a good support team, customizable and easy to use.
Medical Devices





Very nice functions to help keeping the overview and know the audit readyness of your (medical device) product like consistency check and many more with very low documentation-wise effort.





Aligned Elements is so helpful to us in enabling containment of the design and development process. The team @ Aligned are also incredibly receptive to our requests for customisation, and also always quick to act whenever we have issues or bugs.
Biotechnology
FAQ
I'm not sure if our QMS is final. Is implementation still worthwhile?
Aligned Elements is built for QMS evolution. When you adjust processes, we configure the software accordingly without re-implementation or extra costs. The QMS development phase is ideal for Aligned Elements because you start immediately with systematic traceability instead of migrating legacy chaos later.
We have no capacity for POC. Should we talk again in 6 months?
Every month with fragmented documentation costs €10,000-€15,000 in wasted engineering time plus compliance risks. Our 2-week POC requires 4-8 hours of your time with maximum ROI insight. Many customers start POCs parallel to daily business with smaller products. Can you afford 6 more months with 25-45% engineering overhead for documentation?
The change effort seems too high. How long does implementation take?
With solid QMS, we configure, train, and implement Aligned Elements in 2 weeks for your first product. Our record: Customer productive 12 days after contract signing. We offer a 3-4 day package for custom configuration to reflect your QMS. Typical user adoption: We help you setup according to your QMS since we understand normal medical device workflows, deliver the user training and your team will be ready to go for productive use.
Our board has other priorities. How can I push Aligned Elements internally?
Aligned Elements sells best bottom-up through engineering/QA initiatives with measurable quick wins. Start a 2-week POC with one product, present results to the board ("Audit prep reduced from 120 to 24 hours"), and quantify ROI. Boards respond to numbers: "€127,000 annual savings" or "30% faster time-to-market". We support you with executive summary decks and ROI calculators.
The pricing is above our budget. Is there flexibility?
We structure pricing per your budget constraints. Options: Pilot phase with reduced users (2-5 users, €6-15K annual), staged rollout licensing (pay only for active products/users), or multi-year deals. We prefer paid POCs: Invest €2-5K for 4 weeks hands-on experience with real data. If the POC doesn't meet expectations, you have clarity for little investment instead of €200K for a tool that doesn't work. If you move forward with buying Aligned Elements, we even deduct the cost of the POC.
We work with Google Docs. Is Aligned Elements still usable?
Aligned Elements integrates natively with Microsoft 365 for Word/Excel workflows. For Google Docs: 1) You need at least one MS 365 license to maintain Word documents in Aligned Elements. 2) You may export data from Aligned Elements and read in Google Docs any time. The MS 365 license investment is minimal compared to compliance risk of fragmented Google Docs workflows without audit trail.
We change design objects after approval. How does Aligned Elements handle this?
Aligned Elements doesn't irreversibly freeze items after review. After review completion, items can be changed, but the system automatically notifies affected users via inconsistency rules about the new, unreviewed iteration. You can generate files with approved review items for your team while the next iteration runs in parallel. Aligned Elements even supports branching and merging. This supports agile development while maintaining compliance audit trails.
Does Aligned Elements support easy configuration?
Yes. Your admin team (quality managers or systems engineers) can adjust workflows, templates, and traceability rules themselves via GUI-based tools - no code or complex scripting. Examples: Add risk categories, change review workflows, create custom reports. Training takes 2-4 hours. You're also welcome you to leave these tasks up to our support. It's part of your support agreement and we will respond quickly.
How does Aligned Elements differ from JAMA, codeBeamer, or Polarion?
Aligned Elements is built exclusively for medical device manufacturers. Key differences: 1) Rapid configuration from a team with med-tech domain knowledge vs. months of setup. 2) Live traceability for requirements-risk-test linking. 3) Integrated consistency checks for medical device compliance rules. 4) Direct founder access vs. tier-1 helpdesk. 5) Transparent pricing for mid-sized MedTech (€5-50K) vs. enterprise-only (€500K+). We at Aligned understand MedTech workflows, whilst our competitor is often a generic software ALM with MedTech add-on.
What happens after purchase? What does onboarding look like?
Post-purchase journey: 1) Kickoff within 48h with technical onboarding plan. 2) Week 1-2: Software configuration per your QMS + first product import + team training. 3) Week 3-4: Hands-on support during first usage, daily check-ins. 4) Month 2-3: Phased rollout to additional products. Support: Direct access to founder team for technical escalations, typical response under 24h.
Start Your Audit Readiness Transformation Today
Book a 20-min demo and see live how Aligned Elements reduces your audit preparation. No generic sales pitches, just a hands-on walkthrough with your actual MDR/FDA workflows.
Book Free Demo





